Cell Line Development

Creating highly productive clonal cell lines is time-consuming, labor-intensive, and costly. Key considerations when choosing a cell line as the expression platform include:
- Selecting single-cell clones that can produce the biologic of interest
- Screening for clones that can stably produce the biologic at high productivity/titer, with high protein quality
- Ensuring successful scale-up with a robust cell
Featured Categories
Our ready-to-use, off-the-shelf cell line systems greatly reduce the time, cost, and risk of developing new cell lines for production. Whether you are producing antibodies, recombinant proteins, vaccines or viral vectors for gene therapies, our cell line platforms can accelerate your upstream progress.
Biopharma manufacturers place strong emphasis on accelerated clone development, with good quality and regulatory-compliant material for pre-clinical trials. One approach for achieving these goals is the use of a Chinese Hamster Ovary (CHO) platform that includes cell lines and optimized media and feed; this allows fast, easy selection and scale-up of clones for manufacturing, typically reducing cell line development time by up to eight weeks.
A second approach is partnering with a contract development and manufacturing organization (CDMO) that possesses the expertise and technologies to fast-track process development. The CDMO can offer a customized approach, such as automated mini-pool selection, expression cassette design, and super-transfection.
Ensuring continued stability, safety, and quality of the cell line
Cell line characterization is required by regulatory agencies to confirm the species origin and history of the cell line, while testing identity, stability, and purity. Characterization includes phenotypic or genotypic identity testing and a broad range of tests for adventitious agents.
Producing high-quality cell banks for development and manufacture
Production of a high-quality master cell bank (MCB) from the original therapeutic-producing cell line requires that cells be passaged regularly while in culture. However, this increases the chance of genetic alteration, contamination, and loss of expression constructs. It is therefore critical to passage the cells as few times as possible. To prevent alteration, cells are expanded and cryopreserved in a stable condition and stored at ultra-low temperatures. At later stages of development and manufacturing, a working cell bank (WCB) is required. A WCB is produced from a single vial of the MCB that has been grown for several passages and cryopreserved.
Measuring the quality of a biological product
The complexities of biological manufacturing, along with product heterogeneity introduced by cellular expression systems, present significant challenges when measuring the quality of a biologic drug. Newer high-resolution analytical techniques for product characterization can address these challenges early on, providing answers to crucial questions such as the structural attributes of a drug, how it functions biologically, and the effects on clinical performance.
Visit our document search for data sheets, certificates and technical documentation.
Workflow

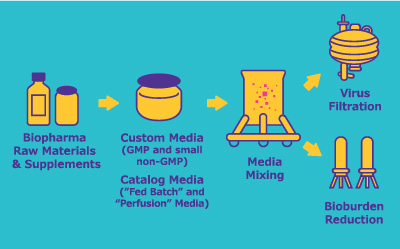
Media and Process Development
Achieving consistent cell culture performance requires careful screening, selection, and optimization of media formulations and feed

Bioreactor Production
Choosing an upstream platform must take many considerations into account, including scalability and quality control of the bioreactors and mixers.

Monoclonal Antibody Manufacturing
Monoclonal antibody manufacturing is a highly templated approach used to produce mAb-based immunotherapies. Robust, scalable process solutions are required at every step to ensure high therapeutic concentration and process safety, while meeting speed-to-market and cost containment concerns.
Related Technical Articles
- Our overview of API solubility and dissolution enhancement methods can help you decide between the many options to take during formulation.
- 3D printing can overcome challenges during formulation development, with a focus on enhancing API bioavailability in solid dispersions. Find out how in this technical article.
- Find out how Hot Melt Extrusion with polyvinyl alcohol (PVA) is used to enhance API solubility.
- In recent years, mannitol has gained popularity as an excipient in solid dosage formulation due to its beneficial physicochemical properties.
- This article describes the advantages of mannitol when used as an excipient in solid drug formulation.
- See All (19)
Related Protocols
- This article describes a purification process resulting in low nanoparticulate impurities sucrose, enabling more stable protein formulations.
- See All (1)
Find More Articles and Protocols
Related Webinars
This webinar will present approaches to the characterization of the physico-chemical and structural attributes in conjunction with understanding the biological activity of monoclonal antibodies, using commercial therapeutic mAbs as case studies.
Learn about challenges faced with traditional expression systems and how a CHO cell line with the glutamine synthetase knock-out via ZFN provides benefits for fast, efficient cell line development with stable and high titer expression.
The results generated by the molecular approach with a better selection of promoters/enhancers combined to a statistical approach including new protocols, mini-pool approach and optimized flow demonstrated that some significant productivity improvements can be achieved using a CHOZn GS -/- and CHOzn K1 expression system.
In this webinar, we present the way we support our customers thanks to advantages provided by the CHOZN® cell line, and a specific strategy for clone selection where semi-automation and pool selection are leveraged, to get it right the first time.
Related Product Resources
- White Paper: Viral Risk Mitigation - A Global Regulatory Perspective
This white paper provides an overview of the global regulatory framework to ensure the viral safety of biologics
- White Paper: Early Product Characterization Mitigates Risks in Biologics Development
Product characterization is the essential foundation for successful biological drug development.
- Tech Brief: Lay the Proper Foundation for Upstream Success
The capabilities and case studies presented within this Tech Brief demonstrate how we combine industry-leading products and customerfocused services to deliver state of the art solutions and support for customers developing innovative therapies.
- Plug & Play Upstream Development Service: BioReliance®Contract Testing
BioReliance® offers testing services for life-changing drugs from pre-clinical to licensed production
How Can We Help
In case of any questions, please submit a customer support request
or talk to our customer service team:
Email custserv@sial.com
or call +1 (800) 244-1173
Additional Support
- Chromatogram Search
Use the Chromatogram Search to identify unknown compounds in your sample.
- Calculators & Apps
Web Toolbox - science research tools and resources for analytical chemistry, life science, chemical synthesis and materials science.
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.
To continue reading please sign in or create an account.
Don't Have An Account?