Sepharose Fast Flow: Purification with Good Resolution and Easy Scale-Up
- Use Sepharose Fast Flow media for purification of proteins.
- Use Sepharose Fast Flow for capture or intermediate purification steps that require good resolution (flows up to 300 cm/h).
- Use a weak ion exchanger such as DEAE, CM or ANX Sepharose Fast Flow, if a strong ion exchanger (substituted with Q, S or SP) does not give the required selectivity.
- Run Sepharose Fast Flow columns on systems such as ÄKTAdesign, FPLC System and HPLC or systems using peristaltic pumps. Appendix 4 gives guidance on how to select the most suitable ÄKTAdesign system.
Sepharose Fast Flow media are based on a matrix of 90 μm particles made from 6% agarose and highly cross-linked for chemical and physical stability. ANX Sepharose 4 Fast Flow (high sub) is based on 4% agarose to form a medium that maintains a high binding capacity when separating large molecules such as thyroglobulin (Mr = 650 000), particularly suitable for large scale production when total binding capacity becomes economically significant.
Sepharose Fast Flow matrices are substituted with a range of ion exchange groups (Q, DEAE, ANX, SP and CM) giving the opportunity to test and use different selectivities (see Chapter 1 for an explanation of strong and weak ion exchangers). Ion exchangers containing strong ion exchange groups (Q and SP) maintain their charge over a broad pH range, allowing selection of the most suitable pH for each application.
Ion exchangers containing weak ion exchange groups (DEAE, CM and ANX) offer alternative selectivities, but over a narrower pH working range. Figure 55 illustrates how the selectivity of Sepharose Fast Flow media changes according to the anion exchange group.
Particle size and bed volumes remain stable, despite changes in ionic strength or pH, to ensure fast separations at high flow rates with good resolution. Methods can be easily scaled up from columns such as HiTrap Q FF (1 mL, prepacked with Q Sepharose Fast Flow) through to large scale columns such as FineLINE. The performance of Sepharose Fast Flow media is well documented and there are many examples of the smooth transfer from the laboratory to pilot scale and on to production.
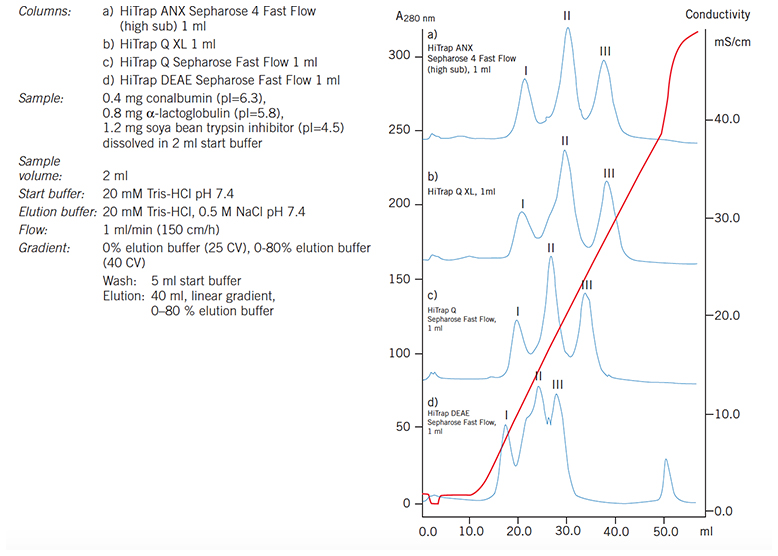
Figure 55.Separation of conalbumin (I), a-lactalbumin (II) and soya bean trypsin inhibitor (III) on a range of anion exchange HiTrap columns demonstrates the difference in selectivity according to the anion exchange group.
Purification options
Figure 56.Sepharose Fast Flow media, with a range of selectivities, are available prepacked in HiTrap and HiPrep columns and in media packs.
*See Appendix 5 to convert linear flow (cm/hour) to volumetric flow rates (mL/min) and vice versa.
**Working pH range refers to the pH interval where the medium binds protein as intended or as needed for elution without adverse long term effects.
***Maximum operating back pressure refers to the pressure above which the medium begins to compress.
- Use prepacked HiTrap columns (1 mL or 5 mL) for media selection, method scouting, group separations, small scale purification, sample concentration or clean-up. Connect up to 3 HiTrap columns in series to scale-up.
- Use prepacked HiPrep columns (20 mL) for method development, group separations, larger scale purification, sample concentration or clean-up. Connect several HiPrep columns in series to increase binding capacity.
Select a production column such as BPG or Chromaflow for larger volumes.
Purification examples
Media scouting
Using 1 mL HiTrap columns the most suitable matrix and charged group for a separation can be quickly and easily selected before optimization and scale-up. In Figure 57 a comparison of elution profiles for the same sample separated under identical conditions on three different media illustrates the differences in selectivity and resolution that can result from changing the charge group and the particle size. The most suitable medium can be selected and conditions optimized according to the requirements for the separation, for example to isolate a single, well-resolved peak or to maximize resolution between several peaks of interest.
Begin by scouting on the strong ion exchangers (Q, S or SP) in order to find the greatest differences in charge between the molecules of interest.
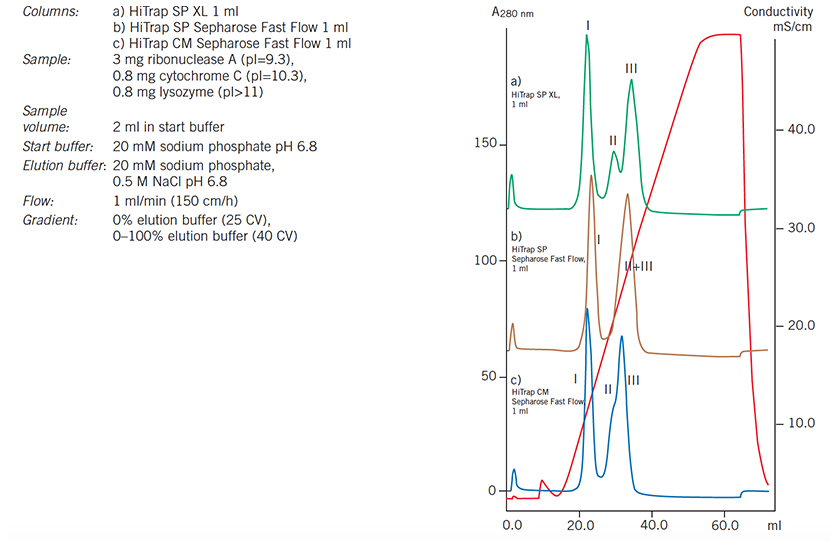
Figure 57.Media scouting: separation of ribonuclease A (I), cytochrome C (II) and lysozyme (III) on HiTrap CM Sepharose Fast Flow 1 mL, HiTrap SP Sepharose Fast Flow 1 mL and HiTrap SP XL 1 mL.
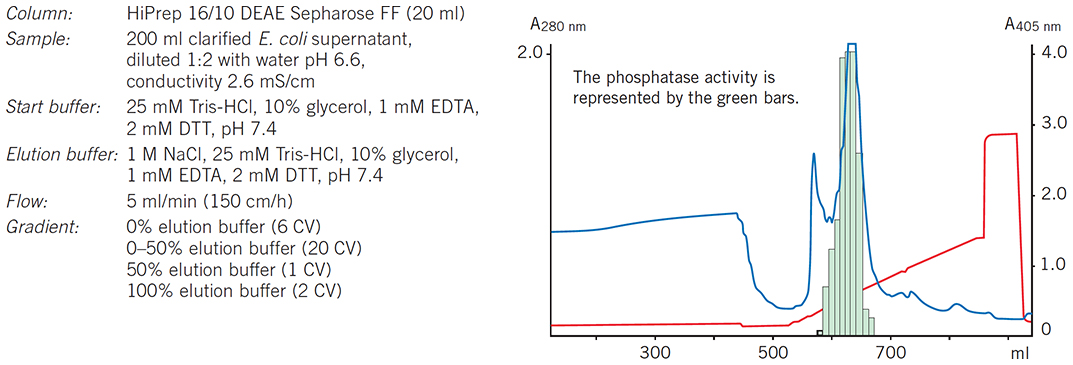
Figure 58.A HiPrep 16/10 DEAE Sepharose Fast Flow column is used as the capture step to concentrate rPhosphatase and remove most of the contaminants.
Scaling-up
Figure 59 shows the ease with which separations can be scaled up on columns prepacked with Sepharose Fast Flow media. Beginning with a 1 mL HiTrap column the reproducibility of the separation has been maintained through a 20-fold scale-up.
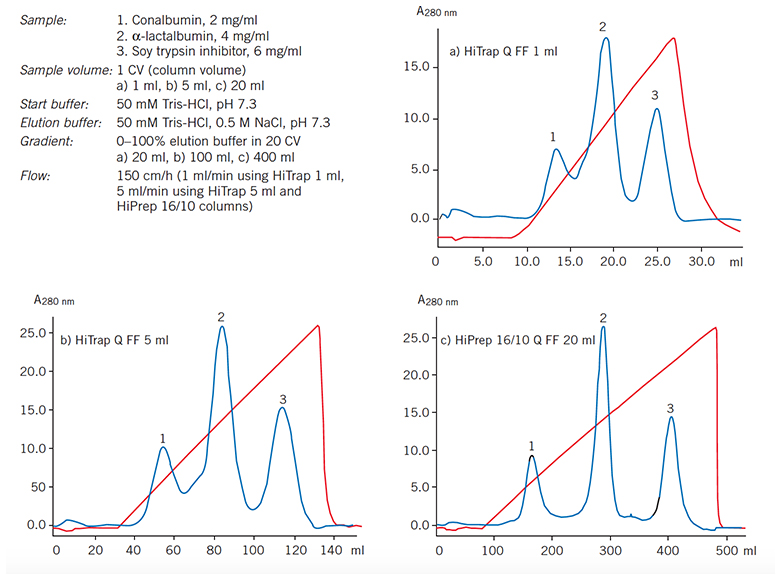
Figure 59.5-fold and 20-fold scale-up using prepacked Q Sepharose Fast Flow columns.
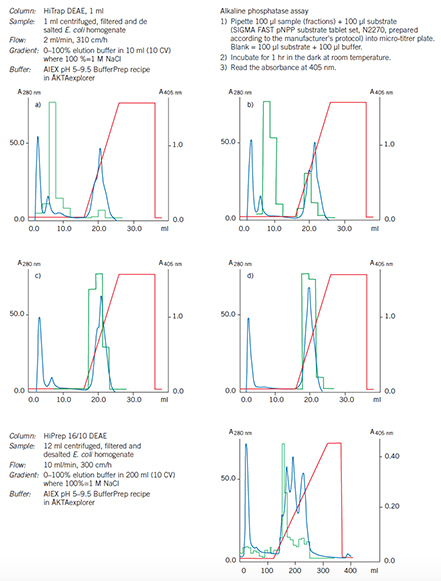
Figure 60.Optimization and scale up on DEAE Sepharose Fast Flow.
Optimizing pH
When the most suitable medium has been selected for a separation, conditions can be optimized further by adjusting parameters such as pH. Figure 61 shows how increasing the pH on a column prepacked with CM Sepharose Fast Flow (HiPrep 16/10 CM FF) significantly improved resolution of a mixture of model proteins.
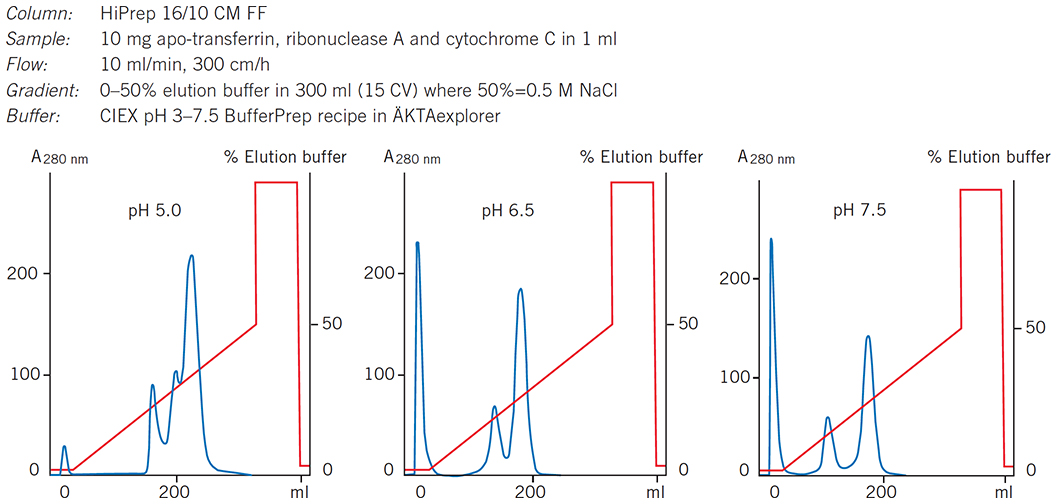
Figure 61.Selecting optimal pH for separation of standard proteins on HiPrep 16/10 CM FF.
Sample concentration
It can be an advantage to concentrate a sample prior to gel filtration in order to minimize sample volume and facilitate a rapid, high resolution size separation. HiTrap columns offer a convenient, ready to use solution for sample concentration. Table 7 on page 89 gives examples of the high concentration factors achieved when concentrating proteins from very dilute starting material using HiTrap columns prepacked with Sepharose HP medium.Similar results can be achieved with HiTrap columns prepacked with Sepharose Fast Flow or Sepharose XL media.
Performing a separation
Guidelines for selection of media, buffer, pH and ionic strength conditions and method optimization are given in Chapter 2. Use the instructions given here as a basis from which to optimize a separation.
- Correct sample and buffer preparation is essential in order to achieve optimal separation and avoid any deterioration in column performance. Samples must be fully dissolved and free from particles or other material likely to interfere with the separation. Refer to Chapter 2 and Appendix 1 for recommendations and advice on sample preparation.
- Filter buffers after all salts and additives have been included. Use high quality water and chemicals. Filter solutions using filters of 1 μm or less. To avoid formation of air bubbles in a packed column, maintain buffers and columns at a constant temperature before and during a run.
- The pH of the start buffer should be at least 0.5–1 pH unit above the pI of the target substance when using an anion exchanger (Q, DEAE or ANX) and 0.5–1 pH unit below the pI of the target substance when using a cation exchanger (SP, CM). See Appendix 2 for recommendations on volatile and non-volatile buffer systems for anion and cation exchangers.
For samples with unknown charge properties, try the following:
- anion exchange (Q)
start buffer: pH 8.0
elution buffer: start buffer including 1 M NaCl, pH 8.0 - cation exchange (SP)
start buffer: pH 6.0
elution buffer: start buffer including 1 M NaCl, pH 6.0
If selectivity is not satisfactory when using a strong ion exchanger (Q or SP), try a weak ion exchanger (DEAE, ANX or CM) instead.
Users of ÄKTAdesign systems with BufferPrep functionality can select one of the buffer recipes recommended for anion exchange chromatography at pH 8 or cation exchange chromatography at pH 6.
First time use or after long term storage
- To remove ethanol, wash with 1 column volume of distilled water at 1 mL/min (HiTrap 1 mL), 5 mL/min (HiTrap 5 mL), 2 mL/min (HiPrep 20 mL), or at 50 cm/h for Sepharose Fast Flow packed in larger columns. This step ensures removal of ethanol and avoids the risk of precipitation if buffer salts were to come into contact with the ethanol. The step can be omitted if precipitation is not likely to be a problem.
- Wash with 5 column volumes of start buffer at 1 mL/min (HiTrap 1 mL), 5 mL/min (HiTrap 5 mL) or 5 mL/min (HiPrep 20 mL).
- Wash with 5 column volumes of elution buffer, same flow as step 2.
- Wash with 5 column volumes of start buffer, same flow as step 2.
- Run a blank elution before applying sample.
Separation by gradient elution
Flow: 1 mL/min (HiTrap 1 mL), 5 mL/min (HiTrap 5 mL), 5 mL/min (HiPrep 20 mL) or at 150 cm/h for Sepharose Fast Flow packed in larger columns. Collect fractions throughout the separation.
- Equilibrate column with 5–10 column volumes of start buffer or until the baseline, eluent pH and conductivity are stable.
- Adjust the sample to the chosen starting pH and ionic strength and apply to the column.
- Wash with 5–10 column volumes of start buffer or until the baseline, eluent pH and conductivity are stable i.e. when all unbound material has washed through the column.
- Begin elution using a gradient volume of 10–20 column volumes and an increasing ionic strength up to 0.5 M NaCl (50%B).
- Wash with 5 column volumes of 1 M NaCl (100%B) to elute any remaining ionically-bound material.
- Re-equilibrate with 5–10 column volumes of start buffer or until eluent pH and conductivity reach the required values.
Separation by step elution
Flow: 1 mL/min (HiTrap 1 mL), 5 mL/min (HiTrap 5 mL), 5 mL/min (HiPrep 20 mL) or at 150 cm/h for Sepharose Fast Flow packed in larger columns. Collect fractions throughout the separation.
- Equilibrate column with 5–10 column volumes of start buffer or until the baseline, eluent pH and conductivity are stable.
- Adjust the sample to the chosen starting pH and ionic strength and apply to the column.
- Wash with 5–10 column volumes of start buffer or until the baseline, eluent pH and conductivity are stable i.e. when all unbound material has washed through the column.
- Elute with 5 column volumes of start buffer + NaCl at chosen ionic strength.
- Repeat step 4 at higher ionic strengths until the target protein(s) has been eluted.
- Wash with 5 column volumes of a high salt solution (1 M NaCl in start buffer) to elute any remaining ionically bound material.
- Re-equilibrate 5–10 column volumes of start buffer or until eluent pH and conductivity reach the required values.
- Save time by using higher flow rates during the high salt wash and re-equilibration steps.
- Do not exceed the maximum recommended flow for the medium.
- If ionic detergents have been used, wash the column with 5 column volumes of distilled water, followed by 2 column volumes 2 M NaCl. Re-equilibrate with at least 10 column volumes of start buffer until the UV baseline, eluent pH and/or conductivity are stable. Organic solvents such as ethanol can be used to remove non-ionic detergents. When selecting an organic solvent, check the chemical stability of the medium to determine a suitable concentration.
- Refer to Chapter 2 for advice on optimizing the separation.
- Check column performance regularly by determining column efficiency and peak symmetry.
- See Appendix 3. Note that this does not apply to HiTrap or HiPrep columns.
Cleaning
Correct preparation of samples and buffers and application of a high salt wash (1 M NaCl) at the end of each separation should keep most columns in good condition. However, reduced performance, a slow flow rate, increasing back pressure or complete blockage are all indications that the medium needs to be cleaned using more stringent procedures in order to remove contaminants.
It is recommended to reverse the direction of flow during column cleaning so that contaminants do not need to pass through the entire length of the column. The number of column volumes and time required for each cleaning step may vary according to the degree of contamination. If the cleaning procedure to remove common contaminants does not restore column performance, change the top filter (when possible) before trying alternative cleaning methods. Care should be taken when changing a filter as this may affect the column packing and interfere with performance.
The following procedure should be satisfactory to remove common contaminants:
- Wash with at least 2 column volumes of 2 M NaCl at 1 mL/min (HiTrap 1 mL), 5 mL/min (HiTrap 5 mL), 5 mL/min (HiPrep 20 mL) or at 40 cm/h with a contact time of 1–2 hours for Sepharose Fast Flow packed in larger columns.
- Wash with at least 4 column volumes of 1 M NaOH (same flow as step 1).
- Wash with at least 2 column volumes of 2 M NaCl (same flow as step 1).
- Rinse with at least 2 column volumes of distilled water (same flow as step 1) until the UV-baseline and the eluent pH are stable.
- Wash with at least 4 column volumes of start buffer or storage buffer (same flow as step 1) until eluent pH and conductivity have reached the required values.
To remove precipitated proteins, lipids, hydrophobically bound proteins or lipoproteins, refer to Appendix 1.
Media characteristics
Composition:
- sulfopropyl (SP), carboxymethyl (CM), quaternary amino (Q) or diethylaminoethyl (DEAE) groups coupled to highly cross-linked 6% agarose via chemically stable ether bonds.
- diethylaminopropyl (ANX) group coupled to highly cross-linked 4% agarose via chemically stable ether bonds.
*Long term pH stability refers to the pH interval where the medium is stable over a long period of time without adverse side effects on the chromatography performance.
Short term pH stability refers to the pH interval for regeneration, cleaning-in-place and sanitization procedures.
All ranges are estimates based on the experience and knowledge within Cytiva.
Chemical stability
For daily use, Sepharose Fast Flow media are stable in all common, aqueous buffers, 1 M NaOH, denaturing agents (8 M urea, 6 M guanidine hydrochloride), with additives such as non-ionic detergents, 70% ethanol, 1 M acetic acid and 30% isopropanol. Sepharose Fast Flow can be used with organic solvents such as dimethylsulfoxide, dimethylformamide, tetrahydrofuran, acetone, chloroform, dichloromethane, dichloroethane and dichloroethane/pyridine (50:50) as well as polar solvents and aqueous/organic isolutions. The water in the medium can be exchanged by the alternative solvent with very little effect on the pore size of the matrix.
Avoid cationic detergents with SP or CM Sepharose Fast Flow. Avoid anionic detergents with Q, DEAE or ANX Sepharose Fast Flow. Avoid oxidizing agents.
Storage
For column storage, wash with 2 column volumes of distilled water followed by 2 column volumes of 20% ethanol. Include 0.2 M sodium acetate in the 20% ethanol solution for SP Sepharose Fast Flow. Degas the ethanol/water mixture thoroughly and apply at a low flow rate to avoid over-pressuring the column. Store at room temperature or, for long periods, store at +4 °C to +8 °C. Ensure that the column is sealed well to avoid drying out. Whenever possible, use the storage and shipping device if supplied by the manufacturer. Store unused media at +4 °C to +30 °C in 20% ethanol. Do not freeze.
- To avoid formation of air bubbles in a packed column, ensure that column and buffers are at the same temperature when preparing for a run.
Materials
To continue reading please sign in or create an account.
Don't Have An Account?