生物反应器生产
不锈钢装置通常成本高昂,耗时长。相比之下,采用一次性使用或混合系统是一条更快、更灵活、资本密集度更低的途径。在选择一次性使用生物反应器平台时,主要的考虑因素包括可扩展性和质量控制:
- 要成功实施大规模一次性使用生物反应器平台,必须证明整个平台与较小尺寸的生物反应器具有同等性能或可扩展性。需要对所有规模的关键工程参数进行表征,包括单位体积的功率输入、混合时间、氧气传输能力和温度映射。
- 要使一次性使用产品和系统达到高质量标准,必须有一个成熟的质量控制系统。该系统需要管理整个生命周期,从组件选择、测试和鉴定,到装配制造、过程中测试和最终产品发布。
特色类别
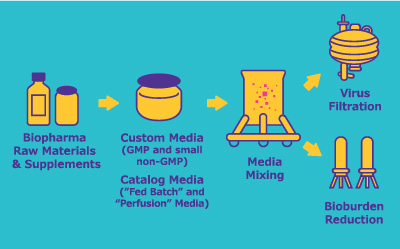
管理上游成套装置的工艺放大和技术转让
从中试规模到工艺规模的转换和技术转让需要精确、无缝的组织、沟通和文档记录。A well-developed technology transfer package:
- Provides a clear understanding of the process and technologies
- Determines the appropriate path to achieve goals of the project
- Aligns with the pharmaceutical quality management system as outlined in ICH Q10
相关文章
- Viral safety in bioprocessing relies on a framework of (1) preventing contamination, (2) detecting contamination, and (3) removing or inactivating viral contaminants.
- 本应用说明描述了Cellvento® 4CHO-X细胞扩增培养基的优势和性能,这款培养基专门开发用于从细胞复苏到N-1阶段的多个细胞扩增步骤,以生成更多生物质接种分批补料生产生物反应器。
- 快速单克隆抗体开发计划可以在开始转染的9个月后交付GMP原料药。我们的mAbExpress™计划具有快速交付、高性价比等优势,非常适合您项目的早期临床研究并全程提供监管支持。
- Multivariate data analysis (MVDA) makes possible a proactive, real-time approach to monitoring, controlling, and predicting quality and productivity in biomanufacturing. The use of proven software with guided PCA and PLS model creation means you don’t need to be a data scientist to explore and analyze your data.
- Key aspects of single-use assembly qualification including quality by design (QbD), quality risk management (QRM) and operator handling and training.
- 查看全部 (21)
相关协议
- This article describes a purification process resulting in low nanoparticulate impurities sucrose, enabling more stable protein formulations.
- 查看全部 (1)
查找更多文章和协议
相关产品资源
- Tech Brief: Mobius® Single-use Bioreactors: Platform Scalability
This brief discusses the characteristics and performance of Mobius® single-use bioreactors, emphasizing their scalability from 3 L to 2000 L for mammalian cell culture and recombinant protein production, and outlines critical engineering parameters for successful scale-up in biomanufacturing processes.
我们能提供哪些帮助
如有任何问题,请提交客户支持请求
或与我们的客户服务团队联系:
发送电子邮件custserv@sial.com
或致电 +1 (800) 244-1173
额外支持
- Chromatogram Search
Use the Chromatogram Search to identify unknown compounds in your sample.
- 计算器与应用_缓冲液计算器_HPLC方法转换计算器-默克生命科学
默克该工具箱包括用于化学、生命科学、材料科学等方面的科学研究工具和资源。
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.
如要继续阅读,请登录或创建帐户。
暂无帐户?