43815
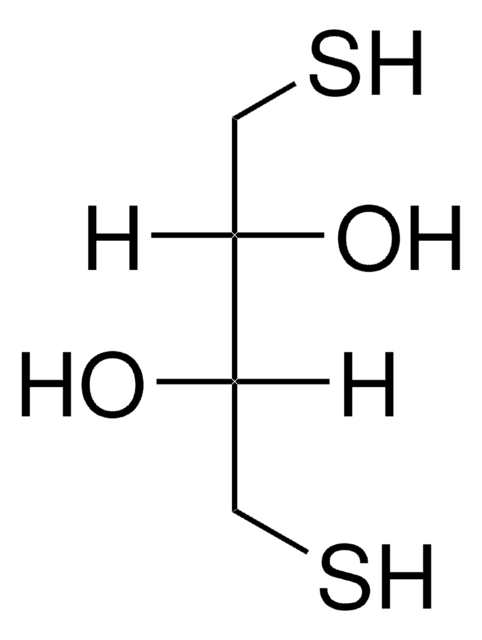
DL-Dithiothreitol
BioUltra, for molecular biology, ≥99.5% (RT)
Recommended Products
grade
for molecular biology
product line
BioUltra
Assay
≥99.5% (RT)
form
powder
reaction suitability
reagent type: reductant
impurities
DNases, none detected
RNases, none detected
insoluble matter, passes filter test
phosphatases, none detected
proteases, none detected
≤0.3% 4,5-dihydroxy-1,2-dithiane
pH
4.0-6.5 (25 °C, 0.1 M in H2O)
mp
41-44 °C (lit.)
solubility
H2O: 0.1 M at 20 °C, clear, colorless
anion traces
sulfate (SO42-): ≤50 mg/kg
cation traces
Al: ≤5 mg/kg
As: ≤0.5 mg/kg
Ba: ≤5 mg/kg
Bi: ≤5 mg/kg
Ca: ≤10 mg/kg
Cd: ≤5 mg/kg
Co: ≤5 mg/kg
Cr: ≤5 mg/kg
Cu: ≤5 mg/kg
Fe: ≤5 mg/kg
K: ≤50 mg/kg
Li: ≤5 mg/kg
Mg: ≤5 mg/kg
Mn: ≤5 mg/kg
Mo: ≤5 mg/kg
Na: ≤500 mg/kg
Ni: ≤5 mg/kg
Pb: ≤5 mg/kg
Sr: ≤5 mg/kg
Zn: ≤5 mg/kg
λ
0.1 M in H2O
UV absorption
λ: 260 nm Amax: 0.400
λ: 280 nm Amax: 0.100
SMILES string
O[C@H](CS)[C@H](O)CS
storage temp.
2-8°C
InChI
1S/C4H10O2S2/c5-3(1-7)4(6)2-8/h3-8H,1-2H2/t3-,4-/m1/s1
InChI key
VHJLVAABSRFDPM-QWWZWVQMSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as an enzyme stabilizer to maintain exebacase stability
- for peptide extraction and protein denaturation
- in sample preparation and reversal of formaldehyde crosslinks before mass spectrometry
- as a buffer component to extract synaptogliosome
Biochem/physiol Actions
Features and Benefits
- BioUltra grade powder suitable for molecular biology
- DNase, RNase, phosphatase, and protease-free insoluble matter, passes the filter test
- Stringently tested and free from trace metals
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Documents related to the products that you have purchased in the past have been gathered in the Document Library for your convenience.
Difficulty Finding Your Product Or Lot/Batch Number?
How to Find the Product Number
Product numbers are combined with Pack Sizes/Quantity when displayed on the website (example: T1503-25G). Please make sure you enter ONLY the product number in the Product Number field (example: T1503).
Example:
Additional examples:
705578-5MG-PW
PL860-CGA/SHF-1EA
MMYOMAG-74K-13
1000309185
enter as 1.000309185)
Having trouble? Feel free to contact Technical Service for assistance.
How to Find a Lot/Batch Number for COA
Lot and Batch Numbers can be found on a product's label following the words 'Lot' or 'Batch'.
Aldrich Products
For a lot number such as TO09019TO, enter it as 09019TO (without the first two letters 'TO').
For a lot number with a filling-code such as 05427ES-021, enter it as 05427ES (without the filling-code '-021').
For a lot number with a filling-code such as STBB0728K9, enter it as STBB0728 without the filling-code 'K9'.
Not Finding What You Are Looking For?
In some cases, a COA may not be available online. If your search was unable to find the COA you can request one.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service